The future of generic medicines isn't just about cheaper pills anymore. It's about smarter ones - combinations that work better, last longer, and help patients stick to their treatment plans. Generic combinations are no longer a niche idea. They're becoming the backbone of how pharma companies stay alive in a market where traditional generics are being priced into oblivion. These aren't your grandfather's copycat drugs. They're engineered versions of older medicines that combine ingredients, tweak delivery methods, or add smart devices to make treatments more effective - without the branded drug price tag.
Think about it: if you're taking three different pills for high blood pressure, diabetes, and cholesterol, wouldn't it be easier to take just one? That's the power of fixed-dose combinations (FDCs). Companies like Teva and Viatris have been building these for years. Instead of just copying a single drug after its patent expires, they're bundling two or more active ingredients into one tablet or capsule. The FDA doesn't just accept this blindly. To get approval, manufacturers must prove the combination works better than taking the drugs separately - not just in lab tests, but in real patient outcomes. That means more clinical data, longer studies, and tougher scrutiny. But the payoff? These products can hold onto 40-60% of their original price for five years after launch. Traditional generics? They lose 80-90% of their value in just two years.
What Makes a Generic Combination "Complex"?
Not all combinations are created equal. The market breaks them into three tiers based on how hard they are to make and test.
- Simple combinations - like oral pills with two active ingredients - make up 62% of today's market. They’re easier to produce and get approved. But growth is slow, around 5.2% per year.
- Complex combinations - think inhalers with built-in dose counters or auto-injectors that deliver two drugs at once - account for 28% of sales. These require precision engineering. The device and drug must work perfectly together. Approval takes longer. But they’re growing at 9.8% a year because they solve real patient problems.
- Super-complex combinations - like nanoparticle systems that target cancer cells or timed-release patches with three active ingredients - are just 10% of the market now. But they’re exploding at 12.7% annual growth. These are the future. They’re expensive to develop, but they command premium prices because there are so few competitors.
The technical hurdles are real. Manufacturing these requires equipment that can mix ingredients within ±2% accuracy. Dissolution tests must match the original branded product within 10% similarity (f2 factor). If the tablet doesn’t break down the same way in your stomach, it won’t work the same way. That’s why so many applications get rejected - not because the active ingredients are wrong, but because the delivery system doesn’t behave like the original.
Regulatory Differences Between the U.S. and Europe
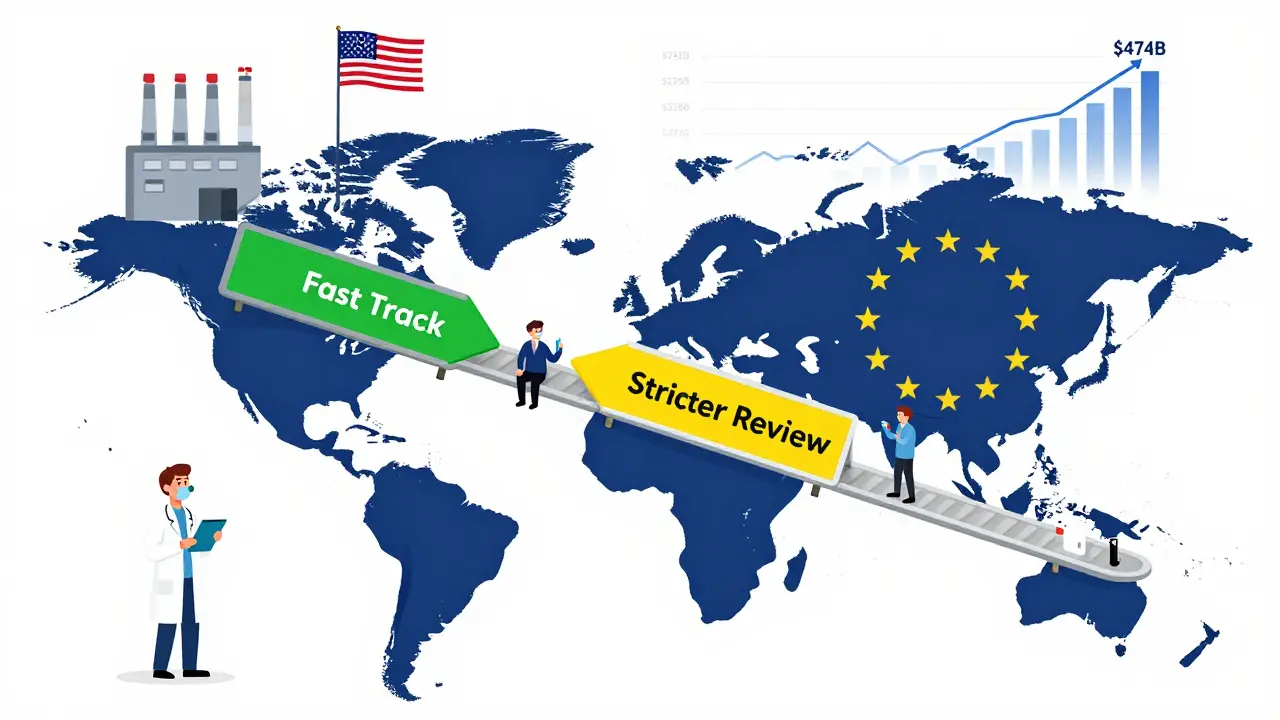
The FDA has been aggressively pushing for faster approvals of complex generics. In October 2025, they launched a pilot program that fast-tracks reviews for products made entirely in the U.S. That’s a big deal. It cuts approval time by 3 to 6 months. Why? Because the U.S. wants to reduce reliance on overseas manufacturing and bring more high-value production home.
Meanwhile, the European Medicines Agency (EMA) is playing it safe. Through Q1 2025, the EU approved only 12 complex generic combinations. The U.S. approved 37. That’s not a coincidence. The EMA demands stricter proof of equivalence, especially for drug-device combos. Companies can’t just say, "It’s the same." They have to show, in detail, how every part of the system - the needle, the spring, the coating - behaves identically to the branded version. This creates a split market. A product approved in the U.S. might still be stuck in review in Europe. That forces companies to build separate development pipelines for each region.
Dr. Janet Woodcock, former head of the FDA’s drug review center, put it plainly: "The FDA is modernizing the ANDA pathway to accommodate increasingly complex products while maintaining scientific rigor." That’s code for: we know these aren’t simple copies anymore, and we’re changing how we evaluate them.

Why This Matters for Patients and Payors
Patients don’t care about regulatory pathways. They care about whether their medicine works, whether they can afford it, and whether they can remember to take it. Generic combinations tackle all three.
Take respiratory disease. Trelegy Ellipta - a branded inhaler combining three drugs - made $2.8 billion in the U.S. in 2024. A generic version isn’t just a copy. It’s a device that delivers the same three drugs, with the same dose counter, same inhalation technique, same reliability. If a generic version hits the market, it could save Medicare and private insurers billions. But it’s not easy to build. The device alone requires 18 months of development just to get the mechanics right.
Same with CNS drugs. Austedo, for movement disorders, hit $1.2 billion in sales. Generic versions are in the works - but they’re not just pills. They’re modified-release capsules that release the drug slowly over 12 hours. That means fewer doses, fewer side effects, better compliance. That’s the value. And it’s why these products can charge 2-3x more than traditional generics while still being cheaper than the brand.
The numbers tell the story: generics make up 90% of U.S. prescriptions but only 20% of spending. That’s unsustainable. Payors want lower costs. Patients want better outcomes. Generic combinations are the only path that satisfies both.
The Big Players Are Betting Everything on This
The industry is consolidating fast. Sandoz split from Novartis to focus purely on complex generics. Viatris and Credence merged for $2.3 billion in 2025 - not to make more simple pills, but to build up their capacity for drug-device combos and extended-release systems. Aspen Pharmacare is pouring millions into "generic semaglutide combinations" - trying to beat the $100 billion GLP-1 weight-loss drug market with a cheaper, combined version.
Device companies are getting in too. Catalent, a leader in drug delivery tech, is partnering with Hikma to build auto-injectors for generic insulin combinations. These aren’t random alliances. They’re strategic moves. You can’t make a working inhaler or auto-injector unless you have deep expertise in both pharma and engineering. That’s why partnerships are becoming the norm.
What’s Next? Three Trends to Watch
- The complexity premium - Products with multiple innovations (combination + extended release + smart device) will command 2-3 times the price of a simple generic. That’s the new normal.
- Regional regulatory gaps - The U.S. will keep pushing faster approvals. Europe will stay cautious. India and China will become the manufacturing hubs, but they’ll struggle to meet U.S. and EU standards unless they invest heavily in quality control.
- Device-pharma partnerships - The companies that win will be those that build bridges between pharma R&D and medical device engineering. No more silos.
The data says it all: the super generics market will grow from $235.6 billion in 2025 to $474.6 billion by 2035. That’s not a prediction. It’s a requirement. Traditional generics are a race to the bottom. Generic combinations are the only way up.
What exactly is a generic combination?
A generic combination is a medicine that combines two or more active ingredients - or a drug with a device - into a single product. Unlike simple generics that copy one drug, these are engineered to improve how the treatment works, such as by making it easier to take, reducing side effects, or delivering the drug more precisely. Examples include fixed-dose pills for blood pressure or inhalers that combine multiple drugs.
Why are generic combinations more expensive to develop than regular generics?
They require much more testing. Simple generics prove they’re bioequivalent to one drug. Generic combinations must prove the whole system works - the drug, the delivery method, the timing, the device - all together. This means more clinical trials, advanced manufacturing controls, and specialized lab tests. Development costs can range from $15 million to $50 million, versus $1-5 million for a standard generic.
Are generic combinations as safe as branded ones?
Yes - if they’re approved properly. The FDA and EMA require proof that the combination performs the same as the original branded product in terms of safety, absorption, and effectiveness. But some experts warn that standards for complex delivery systems are still evolving. The biggest risks come from products that cut corners on device testing, not the active ingredients themselves.
Why is the U.S. market leading in generic combinations?
The U.S. has a combination of factors: strong reimbursement systems, high drug prices that make innovation profitable, a regulatory agency (FDA) actively adapting its review process, and a large patient base with complex chronic conditions. Europe and Asia are catching up, but the U.S. remains the primary testing ground and market for these advanced products.
What’s the biggest threat to the future of generic combinations?
Pricing pressure. Even though these products command higher prices, payers are pushing back hard. If insurers or government programs refuse to cover them at a premium, manufacturers won’t recoup their R&D costs. Without financial incentive, innovation will slow. The key is proving long-term value - fewer hospital visits, better adherence, lower overall care costs - not just lower pill price.
12 Comments
Donna Zurick
March 5, 2026 AT 22:00 PM
This is why I’m bullish on generics
We’re not just making cheaper pills
We’re redesigning treatment itself
Patients don’t want to think about their meds
They just want them to work
And now they can
One click one pill one less thing to worry about
That’s power
Jessica Chaloux
March 7, 2026 AT 02:32 AM
I’m so emotional about this
My grandma took 7 pills a day
Now my cousin takes ONE
And she actually remembers
😭
Mariah Carle
March 8, 2026 AT 04:41 AM
Is this not just capitalism repackaging necessity as innovation
We are solving a problem we created
By fragmenting care into 3 separate prescriptions
Then monetizing the solution
But still
It works
And maybe that’s the point
Philosophy doesn’t fill prescriptions
But better outcomes do
🤔
John Smith
March 8, 2026 AT 09:40 AM
Let’s cut the BS
The FDA isn’t being progressive
They’re desperate
US drug prices are a national embarrassment
So now they’re greenlighting fancy generics to pretend we’re innovating
Meanwhile the real problem
Pharma monopolies
Patent trolling
Insurance middlemen
They’re still untouched
This combo stuff is a shiny distraction
But hey if it helps grandma take her meds
I’ll take it
Just don’t call it progress
Sharon Lammas
March 10, 2026 AT 06:49 AM
I’ve seen too many patients give up because the regimen was too complicated
One pill that works
One device that doesn’t fail
One system that understands human limits
That’s not just science
That’s dignity
And we’re finally building it
marjorie arsenault
March 10, 2026 AT 07:46 AM
If you’re a patient
Take a moment
Think about how many times you’ve forgotten a pill
Or mixed up the schedule
Or been scared by side effects
Now imagine one thing that fixes all of it
That’s what this is
It’s not fancy
It’s not flashy
It’s just better
And that’s enough
Darren Torpey
March 10, 2026 AT 13:43 PM
Generic combinations are the phoenix rising from the ashes of the old generic model
Remember when generics were just white pills with no branding
Now they’re smart devices
Engineered systems
Microfluidic magic
And yes
The price tag reflects the R&D
But so what
It’s still 70% cheaper than the brand
That’s not greed
That’s evolution
Justin Rodriguez
March 10, 2026 AT 18:16 PM
Manufacturing tolerances matter
±2% accuracy isn’t just a number
It’s the difference between a pill that works and one that does nothing
The dissolution f2 factor
10% similarity
That’s the line
Too far
And you get subtherapeutic doses
Too close
And the cost skyrockets
This isn’t copying
This is precision engineering
And it’s why so many applications get rejected
Megan Nayak
March 12, 2026 AT 02:52 AM
Let me guess
The next thing we’ll be told is that we need to pay extra for "premium generics"
Because now they have a "smart dose counter"
And a "patented delivery mechanism"
And a "proprietary coating"
Oh and it’s made in the USA
So now it’s worth 3x more
But still cheaper than the brand
Right
Right
Right
And the next thing
Is that we’ll be told this is "fair"
Because innovation
Meanwhile
Real innovation
Like insulin
Is still priced like a luxury yacht
So yeah
Let’s celebrate this
While the system stays broken
Siri Elena
March 12, 2026 AT 17:09 PM
Oh wow
So now we’re calling complex drug-device combos "generic"
That’s like calling a Tesla a "generic car" because it has four wheels
And then charging $15,000 for it
Because it’s "complex"
How charming
So the brand gets $1000
The generic gets $300
And we call that affordability
How poetic
How very 2025
Pankaj Gupta
March 13, 2026 AT 11:37 AM
The regulatory divergence between FDA and EMA is predictable
The U.S. prioritizes speed to market
Europe prioritizes uniformity and caution
Both have merit
But the real challenge is harmonization
Manufacturers should not need two separate regulatory pathways
Global standards are possible
They just require political will
And investment
India and China can lead this
If they upgrade their quality infrastructure
Not just as suppliers
But as co-architects of global health standards





Shivam Pawa
March 4, 2026 AT 17:17 PM
Generic combinations are the quiet revolution nobody talks about but everyone benefits from
India’s manufacturing base is already pivoting to handle complex delivery systems
The real win isn’t price-it’s adherence
One pill instead of three means fewer missed doses
Chronic disease management becomes feasible when the system is designed for human behavior
Device integration is the next frontier
Not just drugs but interfaces
Think dose counters that sync to phones
Or auto-injectors that log usage
That’s where the value is
Not in cutting cost but in reducing systemic burden
The FDA’s pilot program is smart
Bringing production home reduces supply chain fragility
And yes the tech is hard
But we’ve done harder